Story
Practical lithium–organic batteries enabled by an n-type conducting polymer
Key takeaway
Scientists have developed a new type of battery using a polymer material that can store and release lithium efficiently, allowing for safer and more reliable batteries across a wide range of temperatures.
Quick Explainer
This study reports the development of a practical lithium-organic battery enabled by an n-type conducting polymer called PBFDO. PBFDO overcomes the limitations of previous organic battery materials by exhibiting excellent mixed ionic and electronic transport, low solubility, and the ability to maintain its n-doped state throughout electrochemical cycling. This allows for the realization of high-mass-loading PBFDO cathodes that deliver high capacity, energy density, and cycling stability without the need for additional conductive additives. The wide operating temperature range and exceptional flexibility of the PBFDO-based batteries also demonstrate their potential for applications in extreme conditions and wearable electronics.
Deep Dive
Technical Deep Dive: Practical Lithium–Organic Batteries Enabled by an n-Type Conducting Polymer
Overview
This study reports the development of a practical lithium-organic battery enabled by an n-type conducting polymer, poly(benzodifurandione) (PBFDO), which exhibits excellent mixed ionic and electronic transport and low solubility. The PBFDO cathode maintains its n-doped state throughout electrochemical cycling, delivering high areal capacity, energy density, and cycling stability.
Problem & Context
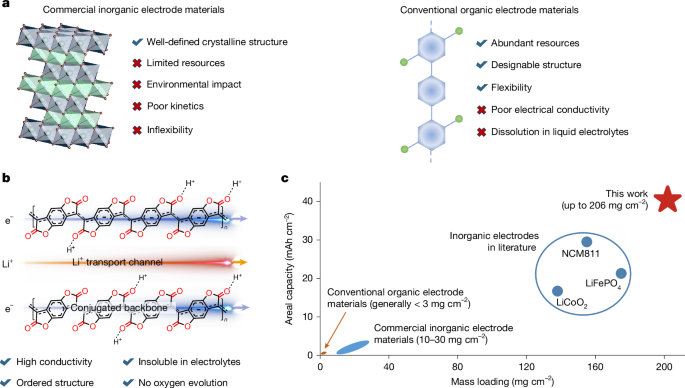
Organic batteries using abundant and recyclable organic electrode materials offer a sustainable alternative to commercial lithium-ion batteries, which rely on resource-limited mineral-derived inorganic electrode materials. However, the practical use of organic batteries has been hindered by the intrinsic insulation and dissolution of organic electrode materials.
Methodology
The researchers developed an n-type conducting polymer, PBFDO, as the cathode material. Key aspects of the methodology include:
- Synthesis and characterization of the ordered, highly conductive PBFDO structure
- Fabrication and electrochemical testing of PBFDO cathodes with high mass loadings up to 206 mg/cm^2
- Assembly and evaluation of practical 2.5 Ah lithium-organic pouch cells
Results
- The PBFDO cathode exhibits stable and reversible redox characteristics, high electrical conductivities, and significant lithium-ion diffusion coefficients, without the need for additional conductive additives.
- Ultrahigh-mass-loading PBFDO cathodes deliver a high areal capacity of 42 mAh/cm^2 and demonstrate robust cycling stability.
- Practical 2.5 Ah lithium-organic pouch cells achieved an impressive energy density of 255 Wh/kg.
- The PBFDO cathode operates efficiently over a wide temperature range from -70°C to 80°C and demonstrates excellent flexibility and safety.
Interpretation
The n-type conducting polymer PBFDO overcomes key limitations of previous organic battery materials, enabling practical lithium-organic batteries with high performance. The stable n-doped state, high conductivity, and low solubility of PBFDO allow for the realization of high-mass-loading cathodes that deliver high capacity and energy density. The wide operating temperature range and exceptional flexibility also mark considerable potential for applications in extreme conditions and wearable electronics.
Limitations & Uncertainties
The source text does not provide details on the specific synthesis and characterization methods for PBFDO, nor does it address potential scalability or cost considerations for manufacturing these lithium-organic batteries.
What Comes Next
Further research may focus on optimizing the PBFDO synthesis and fabrication processes, exploring the long-term cycling stability, and investigating the suitability of PBFDO-based batteries for various practical applications.
Sources: [1] Nature News article: "Practical lithium–organic batteries enabled by an n-type conducting polymer"