Story
Hydrofluorocarbon electrolytes for energy-dense and low-temperature batteries
Key takeaway
Researchers developed a new type of electrolyte that allows lithium-metal batteries to store more energy and operate in colder temperatures, potentially enabling longer-lasting and more capable portable electronics and electric vehicles.
Quick Explainer
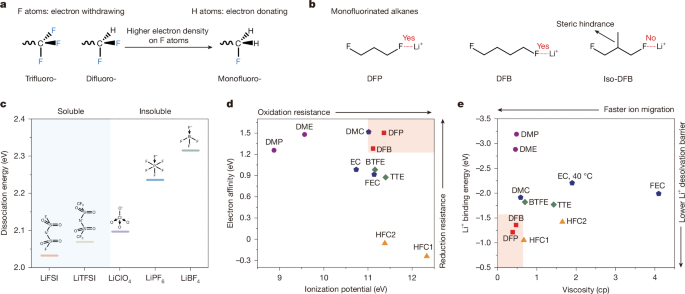
The core idea behind this work is the design of novel hydrofluorocarbon (HFC) solvents as electrolytes for high-performance lithium-metal batteries. The key innovation is the strategic incorporation of fluorine atoms into the solvent structure, which enables weak F-Li+ coordination. This weak coordination facilitates efficient lithium plating and stripping at the electrode interface, even at low temperatures. As a result, the HFC electrolytes demonstrate outstanding properties, including high energy density, low viscosity, and impressive ionic conductivity, enabling batteries that can operate effectively across a broad temperature range.
Deep Dive
Technical Deep Dive: Hydrofluorocarbon Electrolytes for Energy-Dense and Low-Temperature Batteries
Overview
This work explores the use of hydrofluorocarbon (HFC) solvents as electrolytes for high-performance lithium-metal batteries. The key findings are:
- HFC electrolytes enable energy densities over 700 Wh/kg at room temperature and 400 Wh/kg at -50°C
- The HFC electrolytes have low viscosity (0.95 cP), high oxidation stability (>4.9V), and ionic conductivity of 0.29 mS/cm at -70°C
- The weak F-Li+ coordination in HFCs facilitates efficient lithium plating/stripping with Coulombic efficiency up to 99.7% and high exchange current density at -50°C
Methodology
- Synthesized alkanes with monofluorinated structures to create HFC electrolyte solvents
- Designed the HFC molecular structure to have steric hindrance and Lewis basicity to enable high salt dissolution (>2 M)
- Extensively characterized the solvation structure, ion transport mechanisms, and electrochemical performance of HFC electrolytes
Results
- The 1,3-difluoropropane (DFP)-based Li-ion electrolyte exhibited excellent properties:
- Low viscosity (0.95 cP)
- High oxidation stability (>4.9 V)
- High ionic conductivity (0.29 mS/cm at -70°C)
- Incorporating F atoms in the first solvation shell enabled weak F-Li+ coordination, which facilitated efficient Li plating/stripping:
- Coulombic efficiency up to 99.7%
- Exchange current density 10x higher than O-Li+ coordination at -50°C
- HFC electrolytes enabled Li-metal pouch cells with:
- Energy density >700 Wh/kg at room temperature
- Energy density ~400 Wh/kg at -50°C
- Electrolyte amount <0.5 g/Ah
Interpretation
The key innovation of this work is the design of HFC electrolyte solvents that enable high-performance lithium-metal batteries, especially at low temperatures. The weak F-Li+ coordination facilitated by the HFC structure is critical, as it overcomes the challenges of traditional O- and N-based electrolyte solvents which suffer from inefficient charge transfer at the electrode interface.
Limitations & Uncertainties
- The paper does not provide a detailed comparison of HFC electrolytes to other leading low-temperature electrolyte solutions, such as liquefied gas or fluorinated ether-based systems.
- The long-term cycling stability and safety of the HFC electrolytes are not extensively evaluated.
- The methodology for synthesizing and purifying the HFC compounds is not described in detail.
What Comes Next
Based on the promising results, further development and optimization of HFC electrolytes could lead to widespread adoption in energy-dense and low-temperature battery applications. Key future work includes:
- Exploring HFC molecular design principles to further enhance performance
- Evaluating long-term cycling stability and safety under realistic operating conditions
- Scaling up HFC synthesis for commercial viability
- Integrating HFC electrolytes with advanced battery materials and cell designs
Sources: Hydrofluorocarbon electrolytes for energy-dense and low-temperature batteries | Nature