Story
Direct conversion from alkenes to alkynes
Key takeaway
Scientists have discovered a new way to directly convert alkenes (molecules with carbon-carbon double bonds) into alkynes (molecules with carbon-carbon triple bonds). This could enable more efficient and environmentally-friendly chemical synthesis.
Quick Explainer
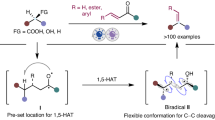
The researchers developed a mild, recyclable selenanthrene-based reagent that can directly convert alkenes into alkynes under relatively gentle conditions. This approach leverages the unique reactivity of the selenanthrene compound to mediate the desaturation of alkenes, enabling the synthesis of desired alkynes while preserving sensitive functional groups. A key advantage is the ability to control the stereochemistry of the final alkyne product, allowing for the inversion or sorting of the alkene Z/E configuration - a capability that is difficult to achieve with existing alkene-to-alkyne conversion methods.
Deep Dive
Direct Conversion from Alkenes to Alkynes
Overview
- Alkynes are widely used as feedstock chemicals and functional groups in organic chemistry, but typical methods for converting alkenes to alkynes use harsh conditions (strong base or high temperatures) that limit their application
- Here, the researchers report a mild, recyclable selenanthrene reagent that mediates the direct conversion of alkenes to alkynes
Methodology
- The researchers developed a selenanthrene-based reagent that can convert alkenes to alkynes under mild conditions
- This method shows broad compatibility with both classical leaving groups and sensitive functional groups, enabling late-stage application in the synthesis of complex alkynes
- The platform also enables Z/E alkene configuration inversion or sorting, which is inaccessible with existing methods
Results
- The selenanthrene reagent effectively converts a wide range of alkenes to the corresponding alkynes under mild conditions
- The method tolerates various functional groups and can be applied late in synthetic sequences to access complex alkyne products
- The reagent enables control over the stereochemistry of the final alkyne product, allowing for inversion or sorting of alkene Z/E configurations
Limitations & Uncertainties
- The source text does not provide detailed mechanistic insights or data on the scope and limitations of the reaction
- It is unclear how the mild conditions and functional group tolerance of this method compare to existing alkene-to-alkyne conversion approaches
What Comes Next
- Further investigation into the reaction mechanism and expanded substrate scope would help elucidate the broader utility and applicability of this selenanthrene-mediated alkene desaturation approach
Sources: [1] Meng, J. et al. Direct conversion from alkenes to alkynes. Nature (2026). https://doi.org/10.1038/s41586-026-10372-3