Key takeaway
Adventitious carbon breaks the symmetry in how oxides exchange charge when touched, important for contact-powered electronics and batteries. This could lead to more efficient and responsive charge storage devices.
Quick Explainer
The key finding is that the presence of adventitious carbonaceous molecules adsorbed on oxide surfaces, rather than just water, is the critical factor that breaks the symmetry in contact electrification between identical oxide samples. Even small differences in the carbonaceous layer coverage can lead to inconsistent charging polarities between sample pairs, explaining the long-standing puzzle. By removing these adsorbates through plasma or mild heating, the researchers were able to restore a consistent charging behavior, demonstrating the central role of the carbon "cocktail" in driving the underlying charge exchange dynamics.
Deep Dive
Technical Deep Dive: Adventitious Carbon Breaks Symmetry in Oxide Contact Electrification
Overview
This work examines the long-standing puzzle of why insulating oxides can exchange electrical charge between samples of the same material, despite the apparent symmetry. The authors show that the key factor is the presence of adventitious carbonaceous molecules adsorbed from the environment, which break this symmetry.
Methodology
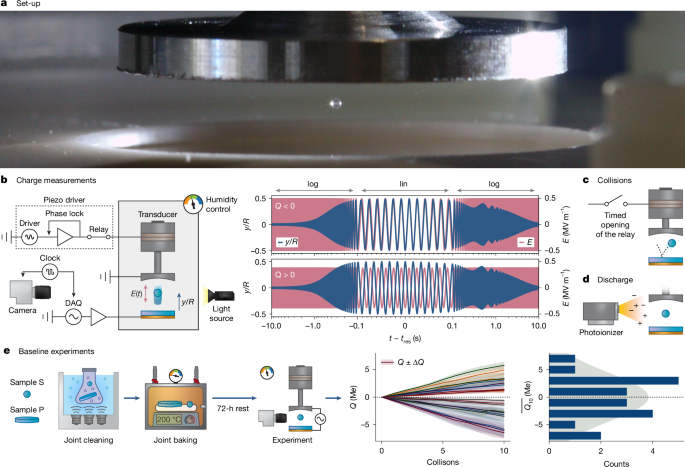
- The researchers used acoustic levitation to study charge exchange between spheres and plates made of identical amorphous silicon dioxide (SiO2).
- They measured the charge acquired by the spheres after collisions with the plates, finding that the charging polarity was random across different sample pairs.
- To test the role of adsorbates, they subjected the samples to plasma treatment or mild baking, which removed adsorbed species.
- After these treatments, the charging polarity became consistent, with spheres charging negatively and plates positively (or vice versa).
Results
- Time-of-flight mass spectrometry revealed that the SiO2 surfaces were covered in a "cocktail" of adventitious carbonaceous molecules, not just water.
- Monitoring the charge exchange and surface composition over time showed that the relaxation dynamics were nearly identical, taking around 10 hours.
- Extending the experiments to other oxide materials (Al2O3, MgAl2O4, ZrO2) revealed that removing the carbon layer could invert the entire triboelectric series.
Interpretation
- The authors conclude that differences in the adventitious carbon coverage between oxide samples, even of the same material, is the key symmetry-breaking factor underlying contact electrification.
- This overturns previous hypotheses that focused on adsorbed water as the critical parameter.
- The history-dependent, out-of-equilibrium nature of carbon adsorption explains why charging polarity can vary randomly between ostensibly identical samples.
Limitations & Uncertainties
- The exact atomic/molecular-scale mechanism by which carbon adsorbates influence charge exchange remains unclear.
- While the authors show carbon is critical, they do not rule out a role for water in modulating charge mobility, as previously proposed.
- The experiments were limited to a small set of oxide materials, so the generality to other insulating solids is uncertain.
What Comes Next
The authors suggest future work should focus on developing theoretical models compatible with the key role of adventitious carbon. Experiments in ultra-high vacuum environments may help isolate the underlying physics, though the authors note that carbon readsorption remains a challenge even in the cleanest conditions.
Sources: