Story
Biosynthesis of cinchona alkaloids
Key takeaway
Researchers uncovered the genes behind the production of cinchona alkaloids, compounds found in anti-malarial drugs. This discovery could lead to improved, more sustainable production of these vital medicines.
Quick Explainer
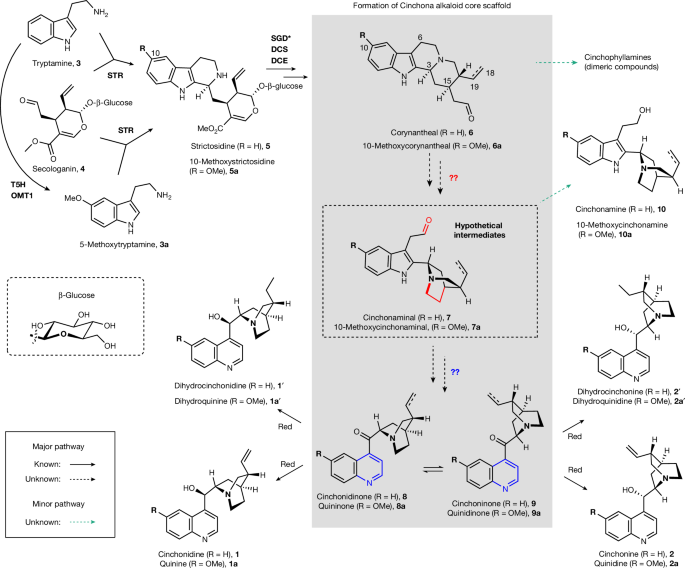
The key innovation in this study was the identification of the previously unknown enzymes and intermediates involved in the biosynthesis of cinchona alkaloids. Researchers discovered that the distinctive quinoline-quinuclidine scaffold is formed through a series of enzymatic steps, including an unexpected cyclization to generate a quaternary amine intermediate, followed by oxidative rearrangement catalyzed by the enzymes cinchonaminal synthase and cinchonamine oxidase. This biosynthetic pathway was then reconstituted in a heterologous host, demonstrating the potential for metabolic engineering approaches to access these medicinally important natural products.
Deep Dive
Technical Deep Dive on Biosynthesis of Cinchona Alkaloids
Overview
Cinchona alkaloids are a group of structurally diverse, nitrogen-containing natural products found in Cinchona plants. These compounds have had a substantial impact in medicine, organic synthesis, and natural product chemistry. Examples include the antimalarial drug quinine, the antiarrhythmic drug quinidine, and the chiral catalysts cinchonidine and cinchonine.
Despite their long history and importance, the biosynthetic mechanism for the formation of the distinctive quinoline-quinuclidine scaffold of cinchona alkaloids has remained largely unknown. This study reports the discovery of the key genes and enzymes responsible for the biosynthesis of this important class of natural products.
Methodology
The researchers employed a multi-pronged approach combining:
- Isotopic labeling experiments
- Gene silencing using virus-induced gene silencing (VIGS)
- Single-nucleus RNA sequencing to map cell-type specific expression
- Comparative transcriptomics across related plant species
- In vitro enzymatic assays
- Heterologous expression in Nicotiana benthamiana
Results
The study uncovered several previously unknown biosynthetic intermediates and enzymes involved in cinchona alkaloid biosynthesis:
- Corynantheol (11) as an on-pathway intermediate: Feeding isotopically labeled corynantheol (11) to Cinchona plants led to the detection of labeled cinchonidine, cinchonine, and cinchonamine, establishing 11 as a key precursor.
- Cinchonium (12) as a quaternary amine intermediate: An unexpected cyclization of 11 was found to generate the quaternary amine compound cinchonium (12), which was then converted to the indole-quinuclidine intermediate cinchonaminal (7).
- Cinchonaminal synthase (CiS) and reductase (CiR): CiS catalyzes the oxidative conversion of 12 to cinchonaminal (7), while CiR reduces 7 to form cinchonamine (10).
- Cinchonamine oxidase (CiO): CiO catalyzes the key oxidative rearrangement of cinchonaminal (7) to form the quinoline scaffold, producing the ketone-quinoline alkaloids cinchonidinone (8) and cinchoninone (9).
- Keto-reductase (KR4): KR4 reduces the ketone of 8 and 9 to form the final cinchona alkaloids.
The researchers were able to reconstitute the entire biosynthetic pathway in Nicotiana benthamiana, producing natural cinchona alkaloids as well as halogenated analogs.
Interpretation
These discoveries uncover the long-standing mystery of how the cinchona alkaloid scaffold is biosynthesized. The identification of the key enzymes involved, such as the unexpected malonylation and demalonylative cyclization steps, reveal new biosynthetic strategies employed by plants.
The ability to produce these alkaloids and analogs heterologously highlights the potential for metabolic engineering approaches to access these medicinally important compounds.
Limitations & Uncertainties
- The mechanism underlying the formation of the C-18–C-19 double bond in the core scaffold remains to be resolved.
- While the keto-reductase KR4 showed clear activity in vitro, it did not exhibit full stereoselectivity, particularly with the methoxylated analogs. An enzyme with higher stereoselectivity may remain to be identified.
- The heterologous expression in N. benthamiana did not fully recapitulate the in planta enzyme activities, likely due to the presence of unknown inhibitory factors.
Future Work
The discoveries from this study open up several avenues for future research:
- Further elucidation of the missing steps and enzymes in the biosynthetic pathway.
- Investigation into the regulation and compartmentalization of the pathway in Cinchona plants.
- Exploration of the full substrate flexibility of the identified enzymes for production of diverse alkaloid analogs.
- Application of these biosynthetic genes for metabolic engineering of cinchona alkaloids in heterologous hosts.